Clinical Data Management – Key Players And Their Duties
Clinical Data Management (CDM) team consists of specialists employed to enter, check and validate the information collected by the survey team. The primary objective is to reduce the number of errors and missing data. Hence, to execute the approach, an appropriate arrangement of the staff members with proper distribution of the work processes and workload is essential so that everything runs smoothly.
Who are the central protagonists in the CDM system?
- Project Supervisor
- Project Clinical Data Manager
- Data Management Assistant
- Database Programmer/Designer
- Database Administrator
- Medical Coder
- Clinical Data Coordinator
- Quality Control Associate
- Data Entry Associate
- Staff responsible for backup or storage
- Administrative support
Roles played by the key players
The CDM is a complex process encompassing multiple roles and responsibilities played by the entire team members. Major roles of some of the prominent players in CDM include:
- Clinical Data Manager
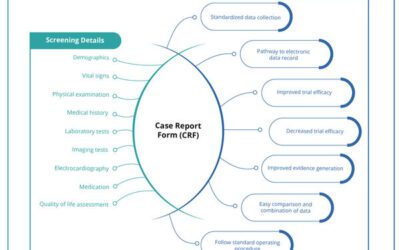
The Clinical Data Manager is responsible for managing the data collected during the clinical study. This encompasses appropriate planning, designing and implementation of the data management plan. Moreover, they have to create case report forms and have to conduct an overview of the data entry, data cleaning, validation of data and reporting.
- Study Monitor
The study monitor is responsible for monitoring the conduction of the clinical study in compliance with the protocol and the Good Clinical Practice Guidelines. This encompasses data collection process monitoring, identifying and resolving any discrepancies or errors in the data entry and ensuring that accurate data is fed into the database.
- Data Management Assistant
The clinical data management assistant has vast major project responsibilities including data cleaning, term coding and includes assistance in maintaining data management files. Moreover, they are also in charge of backups.
- Biostatistician
Responsible for appropriate design and implementation of the statistical analysis plan, data analysis and result interpretation.
- Data entry clerk
The data entry clerk applies different exclusive software tools for entering the data into the database. They have the responsibility that correct data is entered promptly into the database.
- Data Quality Assurance staff
They are responsible for monitoring the data collection process and pointing out any errors or inconsistencies in data. Moreover, they are also responsible for accurate data entry into database swiftly.
All the above responsibilities when coordinated together ensure that data gathered during the clinical study is complete, accurate, and reliable and is readily available for analysis and reporting according to the study protocol.
Skills and qualifications
Clinical Data management department members should possess the following criteria:
- Need to be methodological
- Strongly focused on details of the study process
- Updated with the current technologies in the regulatory field
- Organizational and management skills
- Critical and analytical thinking
- Able to follow instructions continually but should raise queries in appropriate concerns
- Willingness to explore and learn
WorkSure® Clinical Data Management team’s potential
WorkSure® provides an expert team of medical practitioners and medical affairs professionals who are engaged in the development of data management and validation plan, screen design of case report forms (CRF), and online and offline database development. Additionally, high-quality CRF tracking and annotation, data collection, entry, validation and management of discrepancy/query are ensured. WorkSure® CDM team is also involved in medical coding and data reconciliation, database locking as well as unlocking. Data extraction and archival, access control and data security along with data analysis is ensured by the CDM professionals.
Protagonists are highly skilled and expertise comprising of project supervisor, project clinical data manager, data management assistant, database programmer/designer, database administrator, medical coder, clinical data coordinator, quality control associate, data entry associate, staff responsible for back-up or storage as well as overall administrative support.