Clinical Trials on Antibody–Drug Conjugates: Key Challenges
Clinical Trials on Antibody–drug conjugates (ADCs) are gaining significant attention as a promising class of targeted anticancer therapies. The journey of ADCs began with Mylotarg, developed by Pfizer, which received its first regulatory approval in 2000. However, concerns related to safety led to its withdrawal a decade later. Its subsequent re-approval reflects the progress made in understanding and improving this therapeutic class. Today, ADCs have evolved considerably, with multiple agents approved and many more under clinical trials.
ADCs are unique in their design, combining three essential components: a monoclonal antibody, a cytotoxic payload, and a linker that connects the two. This structure enables targeted delivery of highly potent drugs directly to cancer cells by recognizing specific antigens on their surface. Unlike conventional chemotherapy, which affects both healthy and cancerous cells, ADCs aim to enhance treatment precision while reducing systemic toxicity.
The effectiveness of an ADC depends heavily on the careful selection and optimization of its components. Once the antibody binds to the target antigen on the tumor cell, the entire complex is internalized. Inside the cell, the linker is cleaved—often triggered by changes in pH or enzymatic activity—releasing the cytotoxic payload. This payload then destroys the cancer cell. In some cases, the drug can diffuse into neighboring cells, producing what is known as the bystander effect, which can be beneficial in tumors with heterogeneous antigen expression. On the other hand, ADCs with non-cleavable linkers tend to act more selectively, limiting their effect to antigen-positive cells.
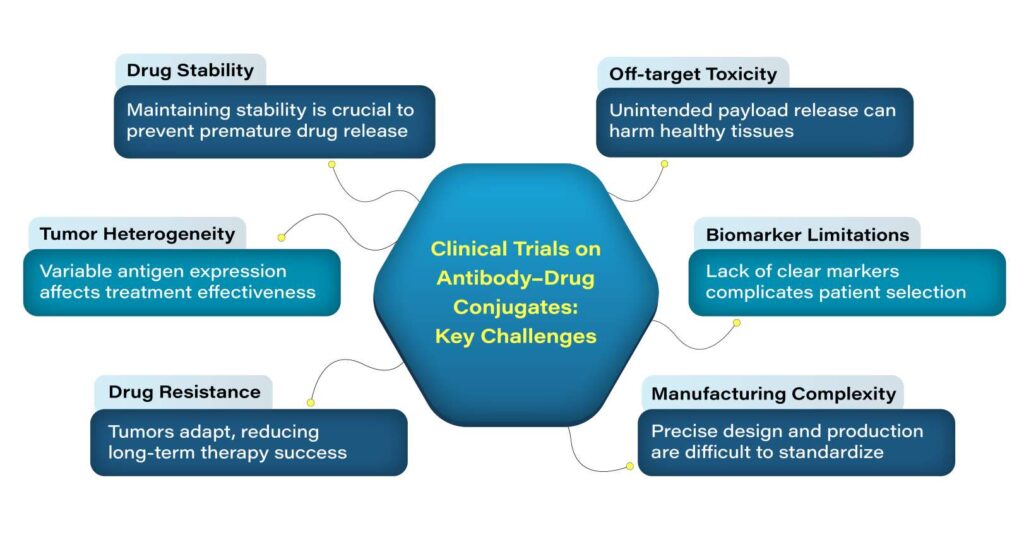
Despite their targeted approach and therapeutic potential, ADCs face several challenges during clinical development. One of the most critical issues is drug stability. For an ADC to be effective, it must remain stable in circulation until it reaches the tumor site. Premature payload release of the cytotoxic drug can lead to off-target toxicity, undermining both safety and efficacy. Additionally, manufacturing instability—such as during conjugation or exposure to environmental conditions like temperature and light—can result in aggregation or loss of activity.
Even when ADCs function as intended, a degree of systemic exposure to the cytotoxic payload is often unavoidable. If the drug is not efficiently cleared, it may lead to cytotoxic payload accumulation in circulation and produce adverse effects similar to those seen with traditional chemotherapy. Managing this balance between efficacy and safety remains a key focus in clinical trials.
Another major challenge lies in patient selection and biomarker identification. While the presence of a target antigen is an important factor, clinical responses have been observed even in patients with relatively low antigen expression. This makes it difficult to define a clear minimum antigen expression threshold for treatment eligibility. Moreover, tumors are not uniform; intratumor variability and variability in antigen levels within and across tumors, along with dynamic tumor biology, can significantly influence therapeutic outcomes and treatment response variability. These factors also complicate patient recruitment in clinical trials and impact clinical trial endpoints.
Key patient-related challenges include:
- Unclear minimum antigen expression threshold required for efficacy
- Variability in antigen levels within and across tumors
- Dynamic tumor biology during treatment
Therapeutic resistance is another important concern. As with many anticancer treatments, tumors can develop mechanisms to evade ADC activity. These may include target antigen downregulation, which reduces drug binding, or increased activity of efflux transporters that actively remove the cytotoxic payload from cancer cells. Given the structural complexity of ADCs, multiple mechanisms of resistance may emerge, making them difficult to predict and overcome.
Despite these limitations, the field of ADCs continues to advance rapidly. Improvements in linker technology, deeper insights into tumor biology, and more refined clinical trial design strategies are helping address many of these challenges. The growing number of approvals and ongoing trials highlights the strong interest and continued investment in this area.
Successfully navigating these challenges requires not only scientific innovation but also robust clinical trial execution. In this context, WorkSure® provides comprehensive support across all phases of clinical development. With expertise in clinical operations, pharmacovigilance, data management, and regulatory compliance, WorkSure® ensures that complex clinical trials—such as those involving ADCs—are conducted efficiently and in alignment with global standards. Our integrated approach helps sponsors manage risks, maintain data integrity, and accelerate the path from research to real-world impact.