Pharmacovigilance and Its Growing Significance in Modern Healthcare
Pharmacovigilance (PV) is defined as the science associated with monitoring and assessing the safety, efficacy and quality of drugs through pre-marketing and post-marketing surveillance. The main focus is to identify adverse effects that may arise from several pharmaceutical products. This process should be conducted throughout the entire lifecycle of the drug development process with special emphasis on any potential safety or efficacy issues that may occur during the course of development or when a drug reaches the market.
Importance of PV
PV is a crucial area with three main objectives: surveillance, operations and focus. Surveillance aims at producing Development Safety Update Reports (DSURs) and Periodic Benefit-Risk Evaluation Reports (PBRER). Operations comprise monitoring of the progress during preclinical studies as well as navigating real-world evidence associated with adverse effects as reported by healthcare professionals or patients. Hence, the focus should be on strong data management systems for appropriate pharmaceutical research protocols maintaining the regulatory guidelines.
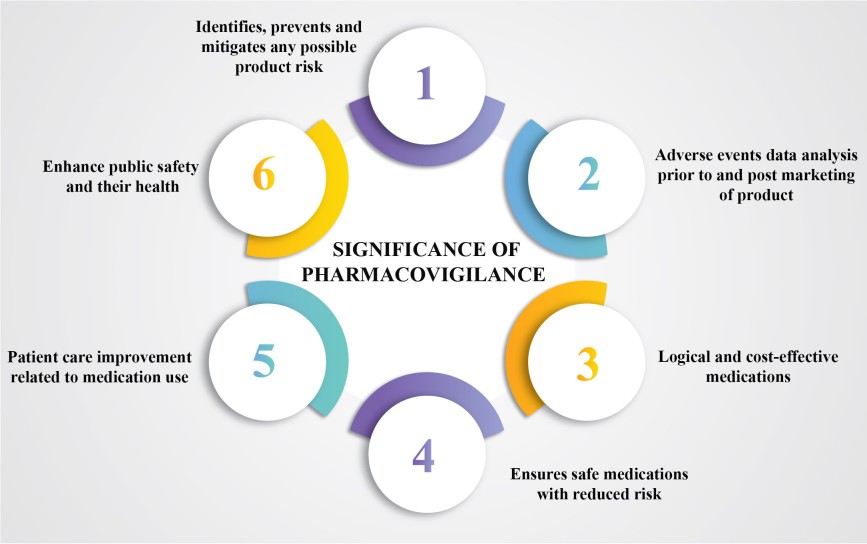
- Identifies, prevents and mitigates any risks present in a product.
- Analysis of adverse events data during pre-clinical trials before marketing of the product as well as post-marketing.
- Drug safety ensures that all medications are safe for use by the consumers while also decreasing the risk associated with their use.
Primary objectives of PV
- Patient care improvement and safety related to medication use and various medical and paramedical procedures.
- Enhance public safety and their health related to medication use.
- Ensure safe, logical and cost-effective medications after proper assessment of benefits, harms, efficacy and associated risks.
- Motivate public awareness of PV with its knowledge, education and medical training.
Role of regulatory authorities on PV
Regulatory Affairs play a major role in the field of PV. Regulatory bodies such as Food and Drug Administration (FDA) provide guidelines associated with the safety and efficacy of a product prior to market approval. Products undergo elaborate repeated testing procedures prior to marketing approval. These include assessing any possibility of side effects or any drug-to-drug interactions. Regulatory bodies review reports of adverse events reported by patients who are on different medications and determine whether action needs to be taken in order to protect patients from harm.
Role of post-marketing surveillance (PMS) on PV
Post-marketing surveillance involves monitoring a product already available in the market and to detect any unanticipated adverse event or other side effect not identified during pre-marketing trials. PMS usually involves healthcare professionals like physicians, pharmacists or nurses who are in close association with the patients taking medications to maintain a long-term safety profile.
Continuous evolving PV in changing clinical environment
PV activities should rigorously evolve continuously as new information is available regarding existing biosimilar products or equivalent new products available in the market. This ensures maximum patient safety during the entire medication period and allowing to effective treatments for their clinical requirements. Special emphasis on pre-clinical trials and post-market surveillance helps pharmaceutical companies to detect any potential adverse effects through monitoring at each stage of development.
Pharmacovigilance Process Solutions at WorkSure®
WorkSure® ensures regular safety status review meetings with stakeholders for the smooth execution of the entire process with reduced overall timelines. Combining our highly skilled team with streamlined processes and innovative tools minimizes redundancies, gaps, and delays at all steps of the Pharmacovigilance processes. We have an experienced team associated with full-time troubleshooting support and integrating applications for signal detection. To name a few, our services in Pharmacovigilance system includes a Pharmacovigilance safety database for drugs/devices/biologics, clinical trial safety management, medical monitoring services, data safety monitoring services, clinical PV auditing services and many others available on the website.