Drug development refers to the process of bringing a new drug molecule into the market through preclinical and clinical practice. It requires substantial investments in terms of capital (∼$2.2 billion), human resources, research skills, and technological expertise. The process of drug development can be broadly classified into preclinical and clinical studies. The […]
All posts tagged: clinical trial
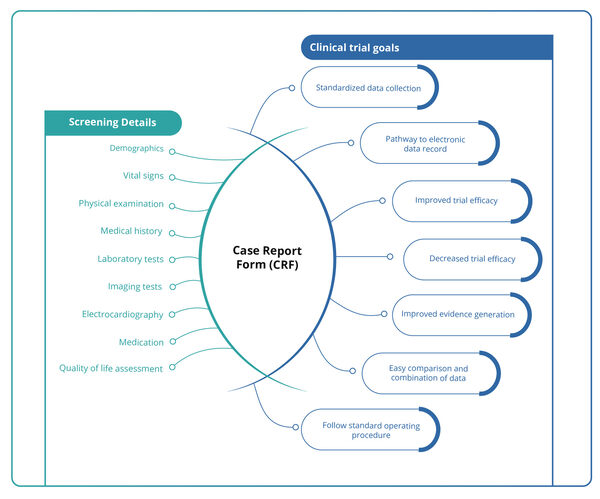
CRF Designing: A challenging step in clinical data management
CRF Designing : Case Report Form (CRF), as defined by the ICH Good Clinical Practice (GCP) guidelines, is a printed, optical, or electronic document used by clinical professionals to accurately capture all protocol-required data for each participant enrolled in a clinical trial. This data is later reported to the sponsor. […]
Clinical Trials: Translating Fundamental Research into Medical Care
Clinical trials are structured biomedical or behavioral research studies on healthy human volunteers as well as patients, intended to answer specific questions about experimental medicine or therapeutic interventions such as new vaccines, drugs, or dietary supplements. These trials are conducted only when the appropriate information is submitted to a government […]
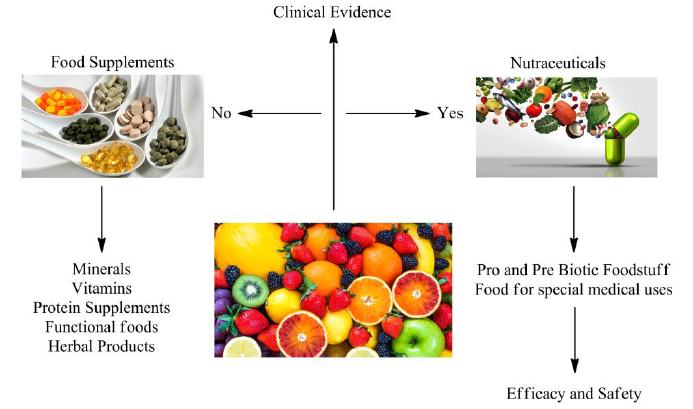
Nutraceuticals in Clinical Trials: A Key to Evaluating Nutraceuticals and Dietary Supplements
“Nutraceuticals,” a term coined by Dr. Stephen DeFelice in 1989, refers to nutritionally enriched foods and dietary supplements that are purified and formulated health products containing bioactive food compounds. These products support optimal health by supplementing the diet with herbs, vitamins, minerals, and amino acids. They are intended to enhance […]
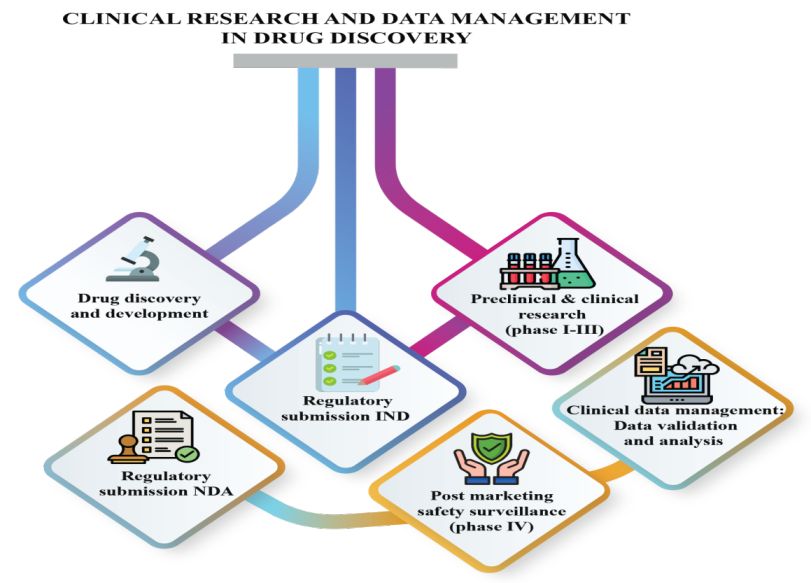
Clinical Research and Data Management: A Comprehensive Approach to Drug Discovery
Clinical research sponsors and pharmaceutical companies encounter numerous obstacles when seeking new drug approvals, often navigating complex regulations and stringent requirements. The clinical research sponsor requires a high-quality data report for getting new drug approval from Food and Drug Administration for their medical products. Clinical research data are important for […]
Medical Affairs Management For Driving Improvement in Patient Outcomes
Medical affairs management identify and address the medical needs associated with products, therapies and disease states thereby driving improvement in patient outcomes in line with commercial objectives. Medical affairs came into existence in pharmaceutical and biotech industries to aid interactions between health care professionals, company representatives and the end-users. Medical […]
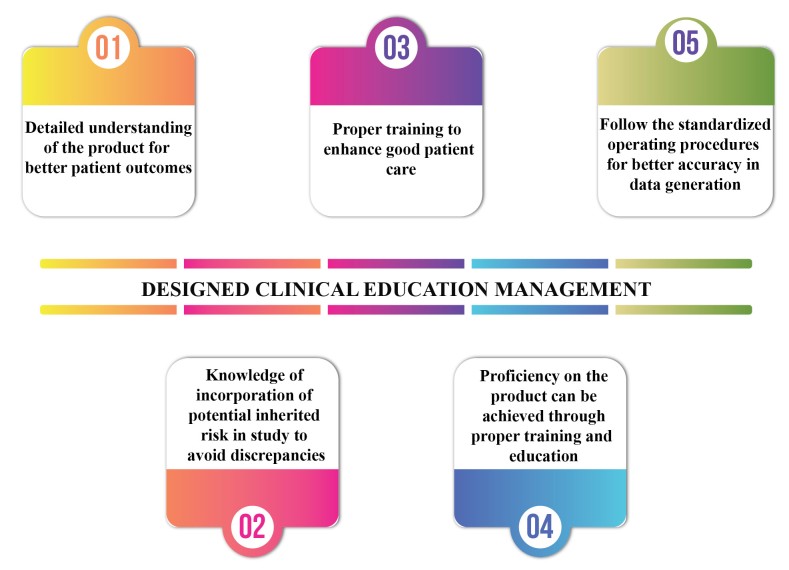
Clinical Education Management for Evolving Clinical Environment
Clinical Education is crucial for healthcare professionals, nurses, pharmacists and physicians for proper maintenance, development and improvement of skills and knowledge regarding the efficiency, comfort and safety of the product. This solves half the equation when it reaches to further enhancement of the delivery of care. However, most often the […]
Clinical Data Management – Key Players And Their Duties
Clinical Data Management (CDM) team consists of specialists employed to enter, check and validate the information collected by the survey team. The primary objective is to reduce the number of errors and missing data. Hence, to execute the approach, an appropriate arrangement of the staff members with proper distribution of […]
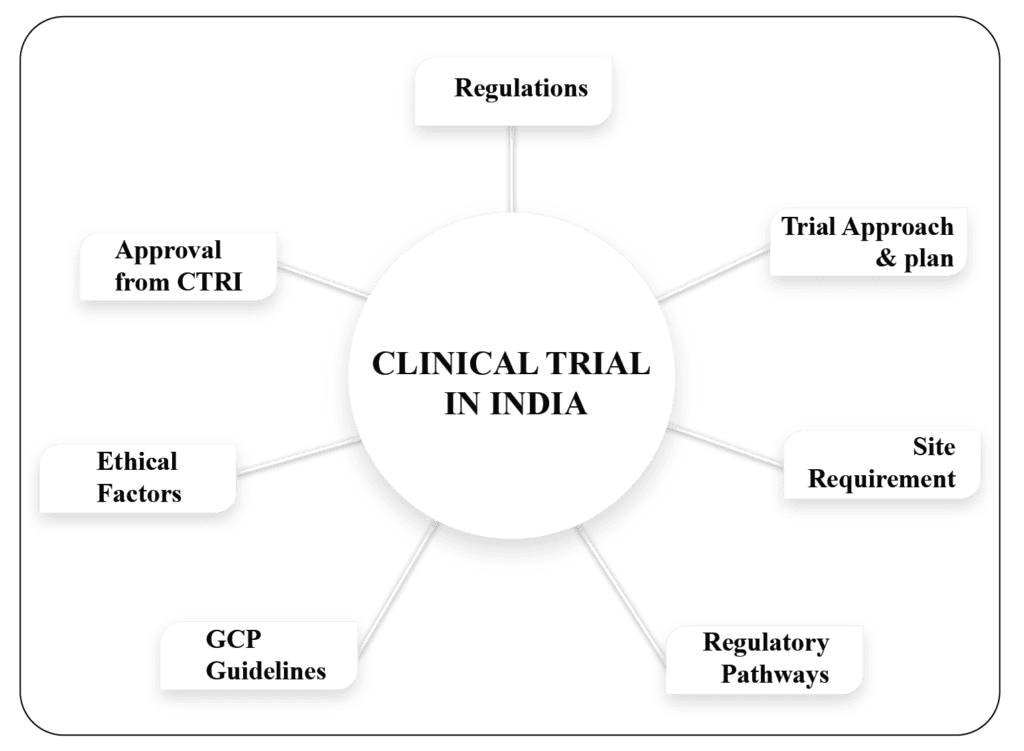
Clinical Trials in India: The Booming Industry in India
Clinical trials are medical research conducted on humans that is planned to assess the efficiency of a drug or treatment and add to medical information. Some trials involve healthy members of the public, and others involve patients who may be offered the option of taking part in a trial during their […]
Clinical Research and Clinical Trials Management
Clinical Research and Clinical Trials Management: Contract research organization, sometimes also called as Clinical research organization (CRO) is an organization that serves pharmaceutical, biotechnology, medical companies and various consumer healthcare industries in the field of clinical research. CRO provides support in new drugs and/or medical devices development that includes pre-clinical […]
Recent Posts
- Zoliflodacin show promise against drug resistant infection urogenital gonorrhoea
- Higher caffeinated coffee intake is significantly associated with lower risk of dementia
- Brain Shunt surgery show promise in treatment of iNPH patients
- Dual creatinine and cystatin C based eGFR measurements can help identify patient at higher health risk
- Orforglipron, Oral GLP-1 drug demonstrates significance weight loss in new clinical trial
Archives
Categories
- Advisory Board Solutions
- Biostatistical Solution for HealthCare
- Clinical Data Management
- Clinical Study
- Clinical Trial
- Education Information Management System
- Electronic Data Management System
- Healthcare Communication
- HEOR
- KOL Management
- Medical Affairs
- Medical Communication
- Medical Writing
- Medico Marketing by WorkSure
- MIMS
- news
- Pharmacovigilance by WorkSure
- Sci-precis Survey Platform
- Uncategorized