Clinical Trials: Translating Fundamental Research into Medical Care
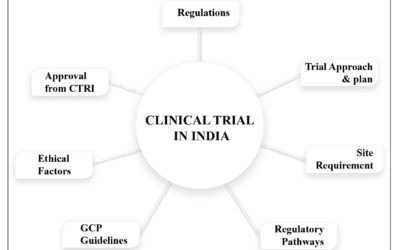
Clinical trials are structured biomedical or behavioral research studies on healthy human volunteers as well as patients, intended to answer specific questions about experimental medicine or therapeutic interventions such as new vaccines, drugs, or dietary supplements. These trials are conducted only when the appropriate information is submitted to a government health authority or ethical committee and receives approval. There are three primary groups involved in conducting clinical trials: sponsors, clinical investigators, and Institutional Review Boards (IRBs). Besides these, human subjects and regulatory agencies are also involved.
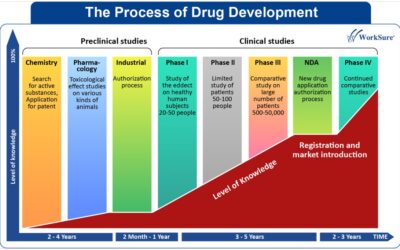
Developing a new medicine is a highly sophisticated and complex task that initially begins with a thorough understanding of a disease or condition. At first, researchers conduct studies in cells, tissues, and animal models to determine whether the disease or condition can be influenced by a new molecule or not. After that, clinical trials establish the new molecule (Investigational New Drug – IND) as a drug and also provide information on how the drug acts on underlying diseases.
For conducting a clinical trial, there must be a proper protocol that describes the aim and objectives of the study. Every clinical trial has its own terms and conditions, i.e., who will take part and who will not. Clinical trials require either volunteers who have the disease in question or healthy volunteers. Ethical guidelines such as the ICH-GCP (International Conference on Harmonisation – Good Clinical Practice) and Good Clinical Practice (GCP) standards serve as global ethical and scientific quality standards for designing, conducting, recording, and reporting clinical trials involving human subjects. Compliance with these standards provides public assurance that the rights, safety, and well-being of trial subjects are protected and consistent with the principles outlined in the Declaration of Helsinki, and that the clinical trial data generated are credible.
Clinical trials are crucial for the drug development process because they enhance our understanding of human physiology and pathophysiology, ultimately translating fundamental research into medical care. Clinical trials are led by a principal investigator, who is usually a medical doctor, along with a research team of nurses and other paramedical staff.
There are different phases of clinical trials:
- Phase I: Testing the drug in a small group of people (nearly 20-80) to assess safety, evaluate appropriate dosage, and detect side effects.
- Phase II: Testing the drug on a larger group (nearly 100-300) to assess its effectiveness and safety.
- Phase III: Testing the drug on large groups (nearly 1,000-3,000) to confirm efficacy, monitor side effects, and collect data to ensure safe usage.
- Phase IV: Post-marketing studies, which include monitoring adverse drug events, assessing risks and benefits, and determining the optimal use of therapy.
If the results of all the required clinical trial phases show that the investigational new drug is safe and effective, the company submits a New Drug Application (NDA) or Biologics License Application (BLA) to the regulatory authority. This application, which includes extensive data from all phases of testing, is a request for regulatory approval to market the new medicine. Scientists at the regulatory authority carefully review all the data and, after evaluating the benefits and risks of the potential medicine, approve the drug for marketing. These clinical trials are conducted through proper regulatory channels to ensure patient safety and compliance with ethical standards.
WorkSure® offers a specialized clinical trial service, providing end-to-end solutions for clinical research and clinical data management. Our expertise spans from developing a robust clinical trial protocol and clinical data management plan to data extraction, statistical analysis, and final reporting. With supportive medical writing services and a commitment to regulatory compliance, WorkSure® ensures seamless execution of clinical trials. With such a comprehensive suite of specialized services, WorkSure® continues to set new standards in clinical research, positioning itself as a leading Clinical Research Organization.
Summary
Developing a new medicine is a complex task. Researchers initially study cells, tissues, and animal models to evaluate the efficacy and mechanism of action of a new molecule. Once clinical trials receive approval, a proper protocol must be followed. Clinical trials require either volunteers who have the disease in question or healthy volunteers. ICH-GCP and Good Clinical Practice (GCP) guidelines must be followed. Compliance with these standards ensures the protection of trial subjects’ rights, safety, and well-being, in line with the Declaration of Helsinki. Four phases of clinical trials must be completed, and if the new drug is found to be safe and effective, the company can submit a New Drug Application (NDA) to the regulatory authority. After thorough review, the regulatory authority may approve the drug for marketing.