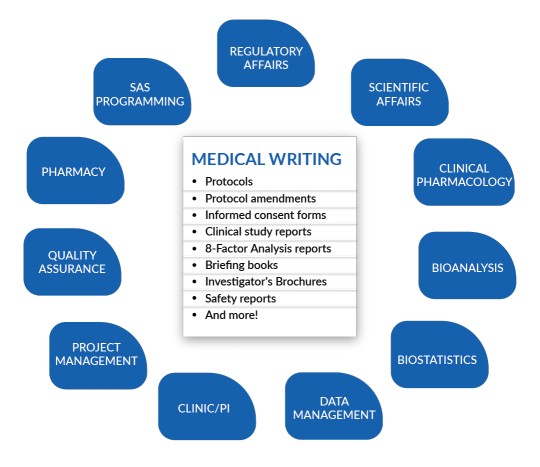
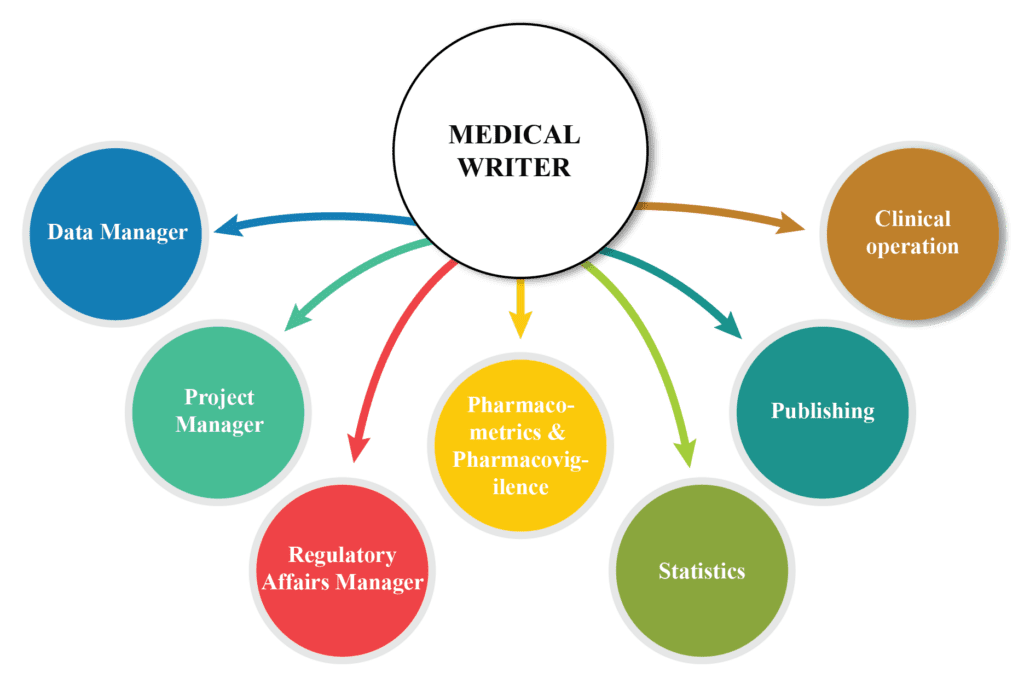
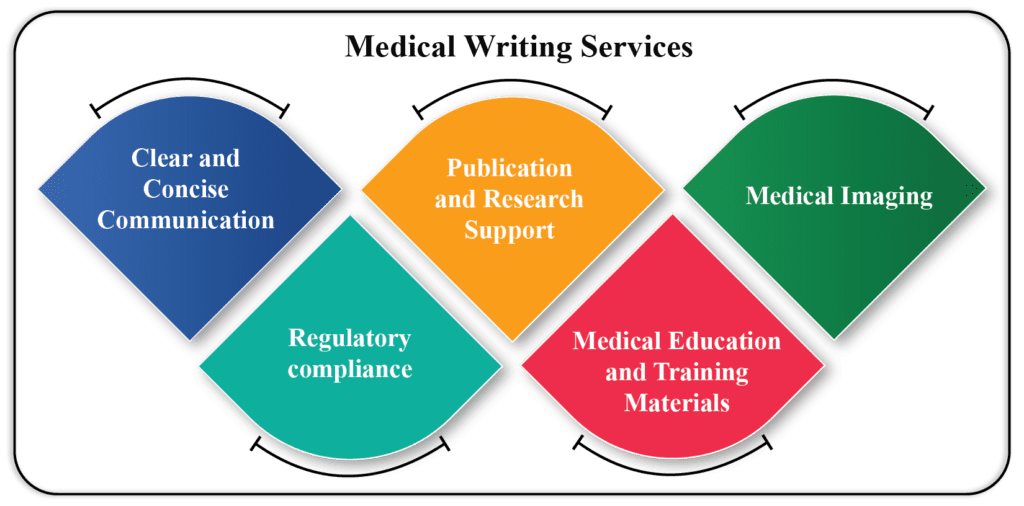
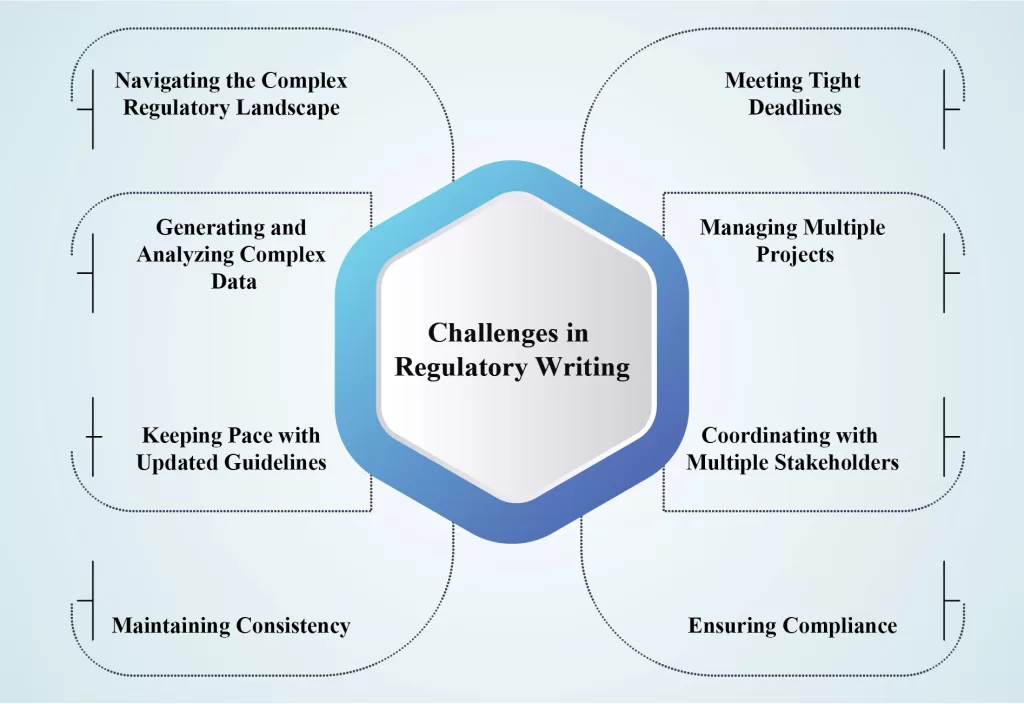
Regulatory Medical writing has globally become an essential tool for generating authenticated documents related to products undergoing clinical trials. The principle of regulatory medical writing is to provide high-quality scientific documentation tailored to the needs of the target audience. Currently, the field has grown more complex due to evolving regulatory […]
All posts tagged: medical writing
Medical Writing: Building Science for Healthcare Community
Medical writing is the process of writing scientific or regulatory documents by professional writers known as medical writers. Medical writers might not be the real scientists who discover the authentic research, but work with the physicians/scientists discovering new fields and thereby assist in representing that data appropriately. The importance of […]
Medical Writing Principles: Scope and Opportunities
Medical writing involves crafting scientific papers by collaborating with doctors and scientists who generate the data, even if the writer isn’t the original researcher. A deep understanding of medical concepts is essential to effectively convey information in a way that the intended audience can understand. Medical writers tailor content to […]
Medical Communication – Importance in Translating Knowledge into Clinical Practice
Medical Communication – Clinical research generates immense knowledge which may guide clinical judgement and decision making when implemented wisely. However, a huge gap exists between the knowledge generation and its use in clinical practice. Therefore, in order to translate knowledge into practice, knowledge generators must ensure an uninterrupted access of […]
Medical Writing : Exploring its Dynamic and Diverse Dimensions
Medical writing is a captivating and rewarding profession that offers numerous opportunities. For those entering the field, it’s natural to have questions about the various types of medical writing and the scope of work involved. Medical writers play crucial roles in various domains, including medical journals, research projects, and medical […]
Medical Writing – Success of Clinical and Regulatory Documents
Medical Writing – Success of Clinical and Regulatory Documents Consistency is a key to planning and is supported by well-designed templates, and style guides that are created by the medical writers to be used throughout the drug development process without dramatic revision. By using style guides for consistent medical writing, […]
Medical Writing Services Effectively Communicate Knowledge and Data in Healthcare
Medical writing is a specialized field that requires extensive knowledge and expertise in writing for medical, pharmaceutical publications. It might target healthcare professionals, regulatory bodies, researchers, sales personnel or may be to educate patients. The work flow for medical writing may vary depending on the type and requirement of document, […]
Medical Writing Regulatory for Product Excellence: Crafting the Path to Success
Medical writing regulatory is the development of various documents intended for submission to Health Authorities (HAs). These documents play a crucial role in conveying essential information accurately, transparently, and clearly to reviewers while adhering to relevant guidelines. Regulatory medical writers hold significant responsibility in the preparation of these documents, ensuring […]
Medical Writing Essentials: Breaking down complex medical concepts
Medical writing involves writing and communicating scientific documents of different types which include regulatory and research-related documents, disease or drug-related educational and promotional literature, publication articles like journal manuscripts and abstracts, content for healthcare websites, health-related magazines or news articles in a clear, brief, plausible, absolute, and convincing manner. The […]
Recent Posts
- Zoliflodacin show promise against drug resistant infection urogenital gonorrhoea
- Higher caffeinated coffee intake is significantly associated with lower risk of dementia
- Brain Shunt surgery show promise in treatment of iNPH patients
- Dual creatinine and cystatin C based eGFR measurements can help identify patient at higher health risk
- Orforglipron, Oral GLP-1 drug demonstrates significance weight loss in new clinical trial
Archives
Categories
- Advisory Board Solutions
- Biostatistical Solution for HealthCare
- Clinical Data Management
- Clinical Study
- Clinical Trial
- Education Information Management System
- Electronic Data Management System
- Healthcare Communication
- HEOR
- KOL Management
- Medical Affairs
- Medical Communication
- Medical Writing
- Medico Marketing by WorkSure
- MIMS
- news
- Pharmacovigilance by WorkSure
- Sci-precis Survey Platform
- Uncategorized