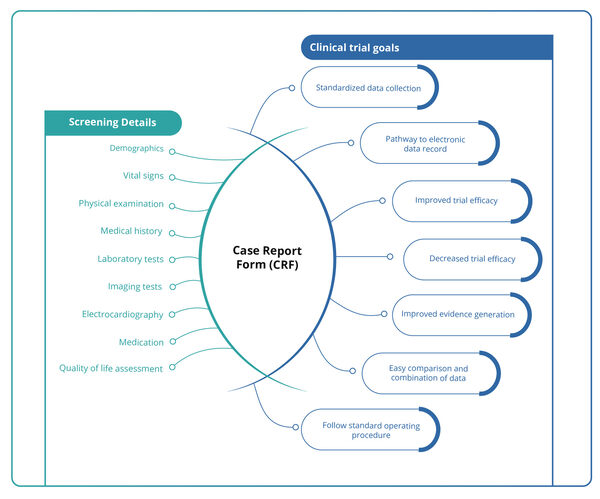
CRF Designing : Case Report Form (CRF), as defined by the ICH Good Clinical Practice (GCP) guidelines, is a printed, optical, or electronic document used by clinical professionals to accurately capture all protocol-required data for each participant enrolled in a clinical trial. This data is later reported to the sponsor. […]
All posts tagged: clinical study
Clinical Data Management – Key Players And Their Duties
Clinical Data Management (CDM) team consists of specialists employed to enter, check and validate the information collected by the survey team. The primary objective is to reduce the number of errors and missing data. Hence, to execute the approach, an appropriate arrangement of the staff members with proper distribution of […]
Clinical Trials in India: The Booming Industry in India
Clinical trials are medical research conducted on humans that is planned to assess the efficiency of a drug or treatment and add to medical information. Some trials involve healthy members of the public, and others involve patients who may be offered the option of taking part in a trial during their […]
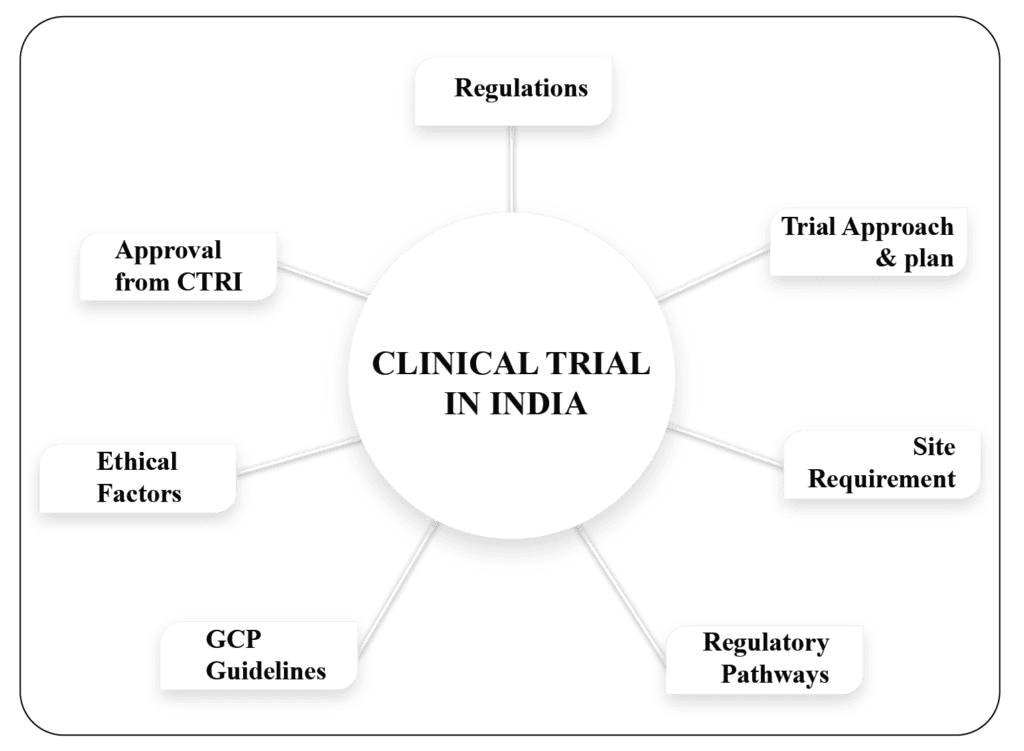
Clinical Trial in India: Exploring Innovative Therapies
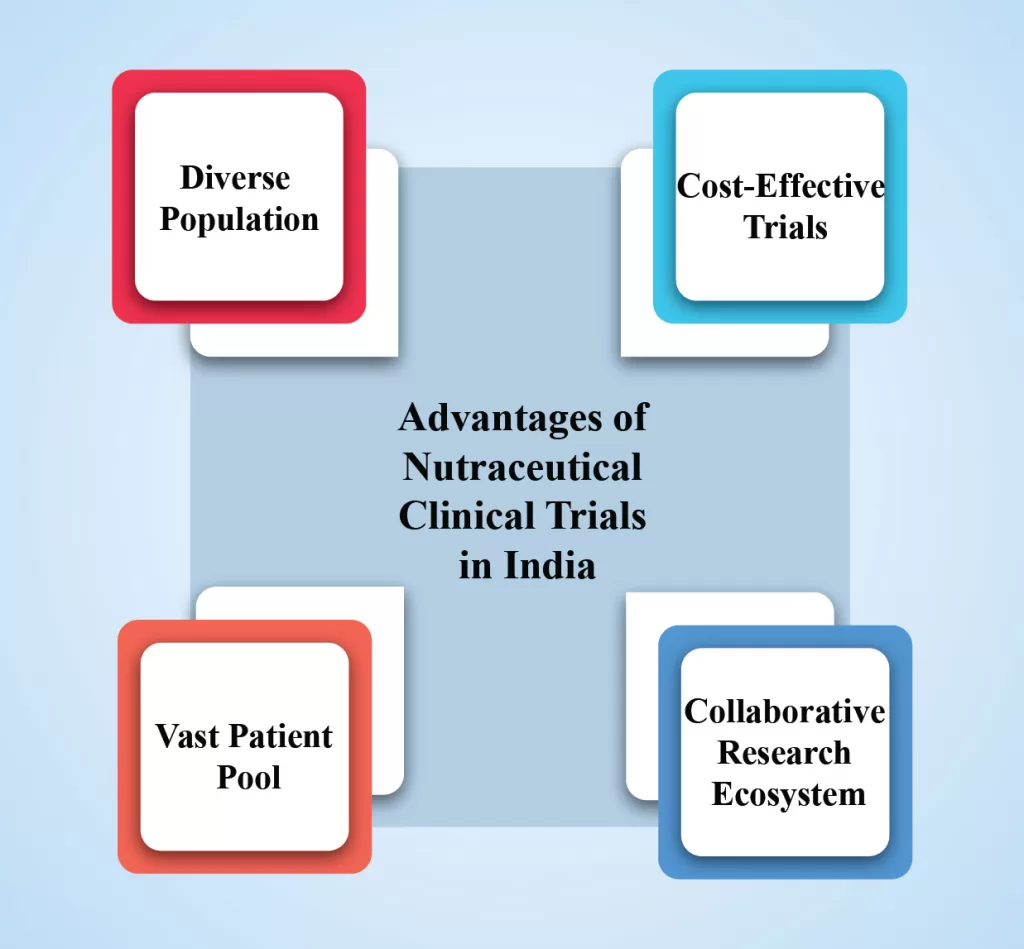
Clinical trial plays a vital role in evaluating the safety and effectiveness of nutraceutical products. Nutraceuticals, the fusion of nutrition and pharmaceuticals, are gaining popularity as natural compounds that offer health benefits and disease prevention. They also provide a crucial avenue for validating these innovative therapies, exploring their potential benefits, […]
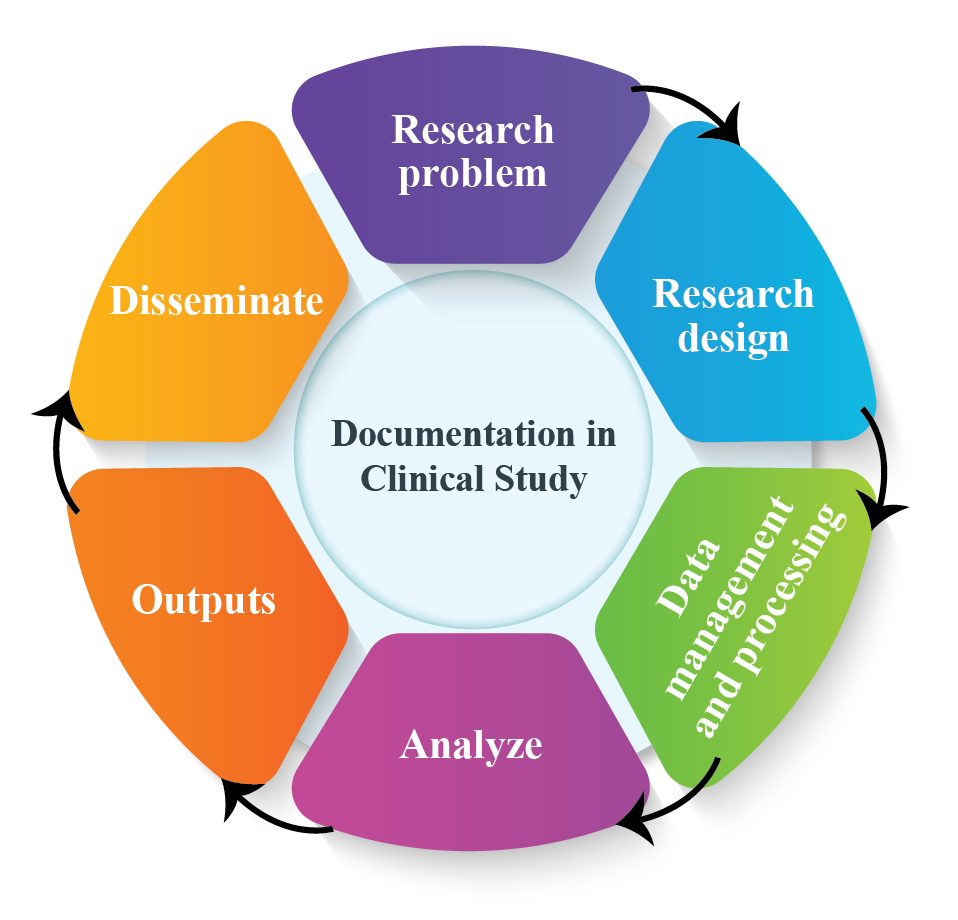
Clinical Study or Scientific Research: Importance of Documentation
Clinical Study: Documentation plays a vital role in clinical study or scientific research. It validates how authentic the research data was collected and verify the result of data. Clinical practice documentation is essential for communication among healthcare providers. It is from this documentation that protocol-specific data are abstracted from and transferred […]
Recent Posts
- Zoliflodacin show promise against drug resistant infection urogenital gonorrhoea
- Higher caffeinated coffee intake is significantly associated with lower risk of dementia
- Brain Shunt surgery show promise in treatment of iNPH patients
- Dual creatinine and cystatin C based eGFR measurements can help identify patient at higher health risk
- Orforglipron, Oral GLP-1 drug demonstrates significance weight loss in new clinical trial
Archives
Categories
- Advisory Board Solutions
- Biostatistical Solution for HealthCare
- Clinical Data Management
- Clinical Study
- Clinical Trial
- Education Information Management System
- Electronic Data Management System
- Healthcare Communication
- HEOR
- KOL Management
- Medical Affairs
- Medical Communication
- Medical Writing
- Medico Marketing by WorkSure
- MIMS
- news
- Pharmacovigilance by WorkSure
- Sci-precis Survey Platform
- Uncategorized