CRF Designing: A challenging step in clinical data management
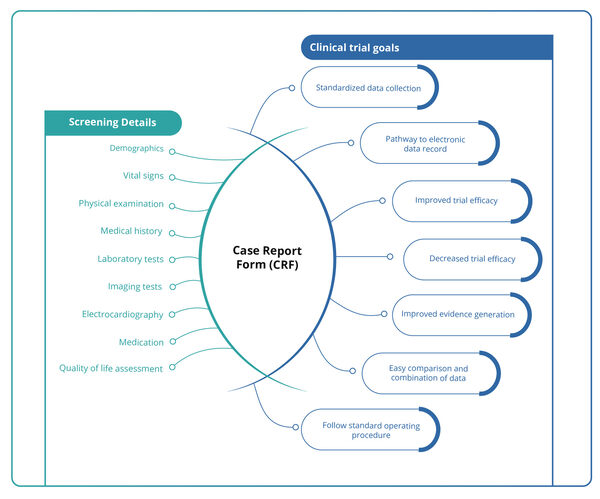
CRF Designing : Case Report Form (CRF), as defined by the ICH Good Clinical Practice (GCP) guidelines, is a printed, optical, or electronic document used by clinical professionals to accurately capture all protocol-required data for each participant enrolled in a clinical trial. This data is later reported to the sponsor. In clinical trials, CRF designing is the most critical step as it represents precise, detailed information that maintains the quality and integrity of data for each subject. The design of the CRF should be study-specific and standardized to meet the requirements of all stakeholders including the investigator, site coordinator, study monitor, data entry personnel, medical coder, and biostatistician.
CRF designing can be classified into two types: Paper CRF (pCRF) and Electronic CRF (eCRF). Nowadays, electronic CRFs (eCRFs) are widely used due to their advantages such as improved data quality, online discrepancy management, and faster database lock. An eCRF reduces the chances of error and avoids data redundancy. Although pCRF is time-consuming and paper-intensive, it is still used in some settings where electronic infrastructure is limited.
During CRF designing, the following points should be noted:
- CRF must be structured, concise, user-friendly, and follow the standard operating procedure (SOP) that ensures high-quality data collection.
- CRF pages should be systematically ordered and aligned with the study protocol.
- The use of fonts, formats, layouts, and queries should be uniform and appropriate.
- The header and footer of the CRF should reflect study trial information.
- Data should be organized in a format that facilitates easy data analysis.
- Data discrepancies should be avoided by maintaining a clear and uncluttered layout.
CRF designing is a very challenging task. The objectives must be clearly defined prior to design, and the data must capture a complete subject profile. A CRF completion manual should be provided to site personnel to promote accurate data entry. These measures will reduce query generation and improve data integrity. It is advisable to maintain a template library of standard CRF modules, as they are time-saving and cost-effective.
Since CRF designing is a crucial step, accuracy in data entry is critical. To overcome challenges, design should be pre-planned and discussed thoroughly with the clinical data management team. To differentiate between various types of responses, different font styles can be used in a well-structured CRF. A query with multiple answers should provide multiple checkboxes to enable easy statistical analysis; pre-coded responses should be used where applicable.
To avoid misinterpretation, CRFs should have a clearly dated format that provides brief but complete information about the patient profile. For best clinical practices, all these elements should be treated as primary considerations when designing a CRF in accordance with SOPs.
Conclusion
By using a user-friendly and study-specific CRF designing, the likelihood of errors and data redundancy is minimized. The CRF should adhere to SOPs to provide accurate subject profiles. CRFs must reflect study-specific and clinically relevant data.
At WorkSure®, we believe in designing concise, structured, and user-friendly CRFs. WorkSure® is equipped to develop CRFs for all phases of clinical trials (Phase I to IV), investigator-initiated studies, hospital/clinic-based healthcare studies, pharmacoeconomic research, and budget impact analyses.
Summary
CRF Designing (Case Report Form) captures patient clinical data. CRFs may be paper-based (pCRF) or electronic (eCRF). eCRFs are increasingly popular due to their accuracy and efficiency, while pCRFs are still used despite being more time-consuming and costly. During CRF design, it is essential to follow SOPs, reduce data redundancy, maintain order, and align with protocol guidelines. An uncluttered layout, proper formatting, and study-specific customization are key. CRFs play a vital role in clinical data collection, making their design a critical task requiring detailed subject profiles and statistical consideration.
WorkSure®‘s trained team—including data managers, biostatisticians, clinicians, and medical writers—designs CRFs in a disease-specific, SOP-compliant, and quality-driven manner.