
Regulatory Affairs

Regulatory affairs services play a pivotal role across various industries, ensuring the safety, efficacy, and compliance of products, particularly in sectors like healthcare, pharmaceuticals, and biotechnology. From the inception of clinical trials to post-marketing activities, regulatory professionals are indispensable in steering companies through the intricate landscape of regulations.
In the pharmaceutical industry, regulatory affairs professionals act as a crucial link between companies and governments globally. Their responsibilities encompass overseeing compliance with diverse regulations, coordinating with global and local regulatory agencies, and advising on regulatory aspects throughout a product's life cycle. They contribute significantly to global drug research, development, and marketing opportunities by understanding the regulatory environment and proposing strategies to minimize market risks. Key areas of their involvement include clinical trial approval, product registrations, regulatory writing, publishing, consulting, and legal representation.
WorkSure® Regulatory Affairs provide crucial guidance on the research and development process, formulation development, product labeling, marketing and secure approvals aligned with clinical research and development standards throughout the product life cycle. Our expertise lies in managing regulatory documentations, regulatory submissions, technical representations, and regulatory approvals in a comprehensive end-to-end approach. WorkSure® Regulatory Affairs services extend even beyond traditional roles by connecting overseas clients with suitable distributors, thereby facilitating effective market penetration and enhanced customer accessibility.
Advantages of WorkSure® Regulatory Affairs Services:
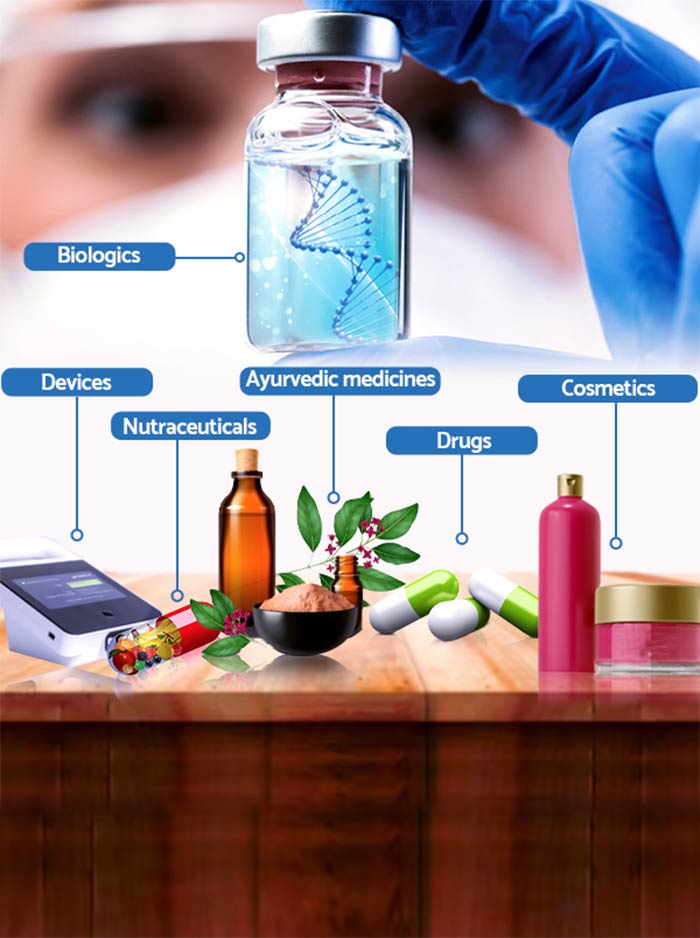
- Provide end-to-end regulatory services for clinical trials involving drugs, devices, biologics, nutraceuticals, ayurvedic, and cosmetic products covering regulatory strategy, documentation, submissions, analysis, approvals, and technical representations
- Deliver expert guidance, advice, and opinions on regional and global regulatory requirements in a comprehensive manner
- Develop customized and cost-efficient regulatory solutions for Clinical Development, Market Authorization, and Post-Marketing activities
- Ensure enhanced Quality Assurance aligned with regional and global regulatory standards (CDSCO, FDA, EMA, MDR, MHRA, PMDA, ICH-GCP Guidelines)
- Facilitate overseas clients by connecting them to the right distributors for effective market penetration and establishment of efficient distribution networks
- Conduct clinical trial audits and maintain ongoing compliance through proactive risk management and staying updated with regulatory changes
- Assist in Ethics Committee Registration support
- Implement Strategic Regulatory Planning by gap assessment and intelligence support
- Offer training on regulatory guidelines and GCP to enhance operational efficiency
- Support in achieving Quality Certifications like ISO Certifications, CE Marking, etc.

WorkSure® Regulatory Affairs Services:
- Comprehensive Regulatory Support for Clinical Trials
- Clinical Trial Audits and Ethics Committee Registration Support
- Tailored Regulatory Solutions throughout Medical Product Life cycle
- Holistic Regulatory Management
- Expert Guidance and Regulatory Monitoring
- Technical/ Medical Regulatory Representations for Queries
- Proactive Risk Management
- Regulatory Trainings to Enhance Operational Proficiency
- Provide Distributer Network and Market Access for Overseas Clients
- Efficient Quality Assurance and Management System
- Quality Certification Support